 Ida Noddack, another German chemist who had worked with Fermi, had earlier proposed that lighter elements could be formed by this type of bombardment. 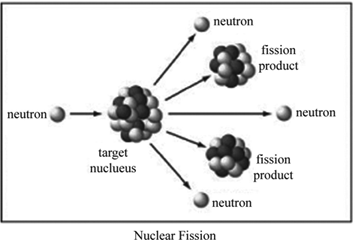
Their work further revealed that the energy released in the bombardment was orders of magnitude greater than any previously known decay. 
Through meticulous radiochemical techniques, Hahn and Strassmann confirmed in 1939 that barium was indeed present among the products, along with many other intermediate-mass nuclei, following the bombardment of uranium by neutrons. See also: Atomic nucleus Coulomb excitation Energy Isotope Nuclear physics PhotonĪlthough this undertaking eventually proved successful, it initially yielded confusing chemical results by seemingly also producing a lighter element, barium ( Z = 56), in very high yields. The development of nuclear physics and the fission process during the twentieth century has played an important role in the technical sector, for instance, with regard to electricity production and medical therapies, as well as affected cultural development and political decision making. More broadly, fission results from disruption of the delicate balance between the attractive nuclear force and the repulsive Coulomb force within a large nucleus and is driven by the fact that nuclear binding energy is maximized for medium-mass nuclei. Fission is a naturally occurring spontaneous decay process of heavy isotopes and can also be induced by the absorption of particles, such as neutrons, protons, or photons, under appropriate conditions ( Fig. The extreme deformation of a very large nucleus followed by splitting of the nucleus into lighter nuclei, predominantly two lighter fragments, in conjunction with a large energy release. Delayed neutron emission by fragments allows control of the chain reactions. Prompt neutron emission during fission provides the capability for a chain reaction of fission events. This probabilistic nature of fission implies that each fission event and its resulting mass and energy distributions are different. Isotopes have an independent fission yield, which is a probability that they will be produced in any given fission event. The liquid-drop model of the nucleus provides an effective qualitative and quantitative explanation of fission.Ī typical fission event releases a total of around 200 million electronvolts (MeV) of energy. The fission process is governed principally by nuclear binding energy and the competition between the attractive nuclear force and the repulsive Coulomb force. This is the principle how fission fragments heat up fuel in the reactor core.A nuclear fission event is the splitting of one nucleus into two or more lighter nuclei fragments. The positive ions and free electrons created by the passage of the charged fission fragment will then reunite, releasing energy in the form of heat (e.g., vibrational energy or rotational energy of atoms). Creation of ion pairs requires energy, which is lost from the kinetic energy of the charged fission fragment causing it to decelerate. The fission fragments interact strongly with the surrounding atoms or molecules traveling at high speed, causing them to ionize. On the other hand most of the energy released by one fission (~170MeV of total ~200MeV) appears as kinetic energy of these fission fragments. Therefore part of the released energy is radiated away from the reactor (See also: Reactor antineutrinos). Most of the fission fragments are highly unstable (radioactive) nuclei and undergo further radioactive decays to stabilize itself. It is much more probable to break up into unequal fragments, and the most probable fragment masses are around mass 95 (Krypton) and 137 (Barium). The average of the fragment atomic mass is about 118, but very few fragments near that average are found. 
Typically, when uranium 235 nucleus undergoes fission, the nucleus splits into two smaller nuclei (triple fission can also rarely occur), along with a few neutrons (the average is 2.43 neutrons per fission by thermal neutron) and release of energy in the form of heat and gamma rays. About 85% of all absorption reactions result in fission. Therefore about 15% of all absorption reactions result in radiative capture of neutrons. The cross-section for radiative capture for thermal neutrons is about 99 barns (for 0.0253 eV neutron). Most absorption reactions result in fission reaction, but a minority results in radiative capture forming 236U. For fast neutrons, its fission cross-section is on the order of barns. Uranium 235 is a fissile isotope, and its fission cross-section for thermal neutrons is about 585 barns (for 0.0253 eV neutron). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
